Catalogue
IgG1 Negative Control, conjugated to PE
Catalog number: GM-4993-CE/IVD| Clone | VI-AP |
| Isotype | IgG1 |
| Product Type |
Negative Control |
| Units | 2ml (100 Tests) |
| Host | Mouse |
| Application |
Direct Immunofluorescence Flow Cytometry Immunofluoresence |
Background
This ready to use Negative Control reagent contains purified, fluorescein or phycoerythrin conjugated mouse immunoglobulin molecules of IgG1 isotype, which have been selected on the basis of their binding characteristics: no specific binding to human cell surface or intracellular antigens, same low range of nonspecific binding to human leukocytes as other Nordic-MUbio Reagents.
This isotype control IgG1 is suitable as a Negative Control to be used in combination with Nordic-MUbio reagents for the:
- Enumeration of Myeloid Cells
- Analysis of Myeloid Differentiation Stage
- Enumeration of B-cells and Precursors
- Enumeration of T-cells and Precursors
- Analysis of Leukemia Cells
- Analysis of Immunodeficiency States
The negative control reagent permits to estimate the degree of non-specific binding of isotype matched immunologbulins to leukocytes via e.g. Fc-receptors. It enables the expert to set flow cytometric parameters accordingly.
Results must be put within the context of other diagnostic tests as well as the clinical history of the patient by a certified professional before final interpretation.
Product
2ml of PE-conjugated VI-AP in PBS pH 7.2, 1% BSA, and 0.05% NaN3, approximately 100 tests.
Product Form: PE
Formulation: PBS pH 7.2, 1% BSA, 0.05% NaN3
Specificity
The clone VI-AP reacts with calf intestine alkaline phosphatase and does not show cross-reactivity with human proteins.
The sensitivity of VI-AP mAb is determined by staining well-defined blood samples from representative donors with serial-fold mAb
dilutions to obtain a titration curve that allows relating the mAb concentration to the percentage of stained cells and geometric MFI
(mean fluorescence intensity). For this purpose, a mAb-concentration range is selected to include both the saturation point (i.e. the mAb dilution expected to bind all epitopes on the target cell) and the detection threshold (i.e. the mAb dilution expected to represent the least amount of mAb needed to detect an identical percentage of cells). In practice, 50 µl of leukocytes containing 10^7 cells/ml are stained with 20 µl mAb of various dilutions to obtain a titration curve and to identify the saturation point and detection threshold. The final concentration of the product is then adjusted to be at least 3-fold above the detection threshold. In addition and to control lot-to-lot variation, the given lot is compared and adjusted to fluorescence standards with defined intensity.
Applications
Direct Immunofluorescence (Staining Procedure) Nordic-MUbio fluorochrome labeled antibodies are designed for use with either whole blood or isolated mononuclear cell (MNC) preparations.
Proposed staining procedure for whole blood in short:
- For each sample add 50 µl of EDTA anti-coagulated blood to a 3-5 ml tube
- Add 20 µl of the appropriate Nordic-MUbio monoclonal antibody conjugate
- Incubate the tube for 15 minutes at 4°C or at room temperature in the dark
- Add 100 µl NM-LYSE (Cat.No. GAS-003) to each tube and incubate for 10 minutes at room temperature
- Add 3-4 ml of destilled water and vortex, incubate for 5-10 minutes at room temperature
- Centrifuge tube for 5 minutes at 300 g - Aspirate supernatant and resuspend pellet in 0.3 ml of sheath fluid
- Analyze immediately or store samples at 2-8° C in the dark and analyze within 24 hours
For “No-Wash” protocol please refer to www.nordicmubio.com
Proposed staining procedure for MNC in short:
- Carefully add 20 µl antibody conjugate and 50-100 µl MNC to the bottom of a tube
- Vortex at low speed for 1-2 seconds
- Incubate for 15-30 minutes at 2-8°C or at room temperature
- Centrifuge tubes for 5 minutes at 300 g
- Remove supernatant, resuspend cells in 2-5 ml of phosphate buffered saline (PBS) and centrifuge cells again for 5 minutes at 300 g
- Remove supernatant and resuspend cells in sheath fluid for immediate analysis or resuspend cells in 0.5 ml 1 % formaldehyde and store them at 2-8°C in the dark. Analyze fixed cells within 24 hours
Storage
Nordic-MUbio monoclonal antibody reagents contain optimal concentrations of affinity-purified antibody. For stability reasons this monoclonal antibody solution contains sodium azide. These reagents should be stored at 2-8°C (DO NOT FREEZE!) and protected from prolonged exposure to light. If a slight precipitation occurs upon storage, this should be removed by centrifugation. It will not affect the performance or the concentration of the product. Stability of the reagent: Please refer to the expiry date printed onto the vial. The use of the reagent after the expiration date is not recommended.
Caution
When used for in vitro diagnostic purposes results must be put within the context of other diagnostic tests as well as the clinical history of the patient by a certified professional before final interpretation. Analyses performed with this antibody should be paralleled by positive and negative controls. If unexpected results are obtained which cannot be attributed to differences in laboratory procedures, please contact us. This product may contain hazardous ingredients. Please refer to the Safety Data Sheets (SDS) for additional information and proper handling procedures. Dispose product remainders according to local regulations.This datasheet is as accurate as reasonably achievable, but our company accepts no liability for any inaccuracies or omissions in this information.
Warranty
The products sold hereunder are warranted only to conform to the quantity and contents stated on the label at the time of delivery to the customer. There are no warranties, expressed or implied, that extend beyond the description on the label of the product. Exalpha`s sole liability is limited to either replacement of the products or refund of the purchase price. Exalpha is not liable for property damage, personal injury, or economic loss caused by the product.
CE Mark
CE
Safety Datasheet(s) for this product:
| NM_Sodium Azide |

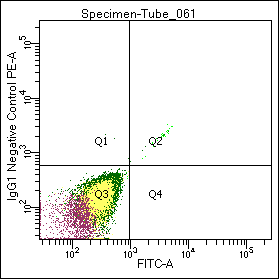
Figure 1. Flow cytometric analysis of normal white blood cells with GM-4993, a PE- labeled negative control IgG preparation.

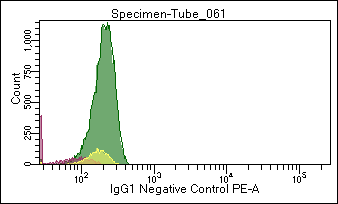
Figure 2. Flow cytometric analysis of normal white blood cells with GM-4993, a PE- labeled negative control IgG preparation.