Catalogue
Mouse anti Basal cell Cytokeratin
Catalog number: MUB0312P| Clone | RCK103 |
| Isotype | IgG1 |
| Product Type |
Monoclonal Antibody Primary Antibodies |
| Units | 0.1 mg |
| Host | Mouse |
| Species Reactivity |
Canine Chicken Guinea Pig Hamster Human Quail Rabbit Rat Swine Zebrafish |
| Application |
Flow Cytometry Immunocytochemistry Immunohistochemistry (frozen) Western Blotting |
Background
Cytokeratins are a subfamily of intermediate filament proteins and are characterized by a remarkable biochemical diversity, represented in Human epithelial tissues by at least 20 different polypeptides. They range in molecular weight between 40 kDa and 68 kDa and isoelectric pH between 4.9 – 7.8. The individual Human Cytokeratins are numbered 1 to 20. The various epithelia in the Human body usually express Cytokeratins which are not only characteristic of the type of epithelium, but also related to the degree of matuRation or differentiation within an epithelium. Cytokeratin subtype expression patterns are used to an increasing extent in the distinction of different types of epithelial malignancies. The Cytokeratin antibodies are not only of assistance in the differential diagnosis of tumors using immunohistochemistry on tissue sections, but are also a useful tool in cytopathology and flow cytometric assays.
Source
RCK103 is a Mouse monoclonal IgG1 antibody derived by fusion of SP2/0-Ag14 Mouse myeloma cells with spleen cells from a BALB/c Mouse immunized with a mix of cell preparations containing Human Cytokeratins.
Product
Each vial contains 100 ul 1 mg/ml purified monoclonal antibody in PBS containing 0.09% sodium azide.
Formulation: Each vial contains 100 ul 1 mg/ml purified monoclonal antibody in PBS containing 0.09% sodium azide.
Specificity
RCK103 is a Cytokeratin antibody recognizing (amongst others) Cytokeratin 5. This monoclonal antibody stains basal cells in combined and stRatified epithelial tissues. It recognizes the stem cell population, including the so-called amplifying cells in the prostate epithelium.
Applications
RCK103 is useful for immunoblotting, immunocytochemistry, immunohistochemistry on frozen tissues and flow cytometry. Optimal antibody dilution should be determined by titration; recommended range is 1:100 – 1:200 for flow cytometry, and for immunohistochemistry with avidin-biotinylated Horseradish peroxidase complex (ABC) as detection reagent, and 1:100 – 1:1000 for immunoblotting applications.
Storage
The antibody is shipped at ambient temperature and may be stored at +4°C. For prolonged storage prepare appropriate aliquots and store at or below -20°C. Prior to use, an aliquot is thawed slowly in the dark at ambient temperature, spun down again and used to prepare working dilutions by adding sterile phosphate buffered saline (PBS, pH 7.2). Repeated thawing and freezing should be avoided. Working dilutions should be stored at +4°C, not refrozen, and preferably used the same day. If a slight precipitation occurs upon storage, this should be removed by centrifugation. It will not affect the performance or the concentration of the product.
Caution
This product is intended FOR RESEARCH USE ONLY, and FOR TESTS IN VITRO, not for use in diagnostic or therapeutic procedures involving humans or animals. It may contain hazardous ingredients. Please refer to the Safety Data Sheets (SDS) for additional information and proper handling procedures. Dispose product remainders according to local regulations.This datasheet is as accurate as reasonably achievable, but our company accepts no liability for any inaccuracies or omissions in this information.
References
1. Feitz, W. F., Debruyne, F. M., Vooijs, G. P., Herman, C. J., and Ramaekers, F. C. (1986). Intermediate filament proteins as tissue specific markers in normal and malignant urological tissues, J Urol 136, 922-31.
2. Verhagen, A. P., Aalders, T. W., Ramaekers, F. C., Debruyne, F. M., and Schalken, J. A. (1988). Differential expression of Keratins in the basal and luminal compartments of Rat prostatic epithelium during degeneRation and regeneration, Prostate 13, 25-38.
3. Schaafsma, H. E., Ramaekers, F. C., van Muijen, G. N., Ooms, E. C., and Ruiter, D. J. (1989). Distribution of Cytokeratin polypeptides in epithelia of the adult Human urinary tract, Histochemistry 91, 151-9.
4. Smedts, F., Ramaekers, F., Robben, H., Pruszczynski, M., van Muijen, G., Lane, B., Leigh, I., and Vooijs, P. (1990). Changing patterns of Keratin expression during progression of cervical intraepithelial neoplasia, Am J Pathol 136, 657-68.
5. Kuijpers, W., Tonnaer, E. L., Peters, T. A., and Ramaekers, F. C. (1991). Expression of intermediate filament proteins in the mature inner ear of the Rat and Guinea Pig, Hear Res 52, 133-46.
6. Vos, J. H., van den Ingh, T. S., de Neijs, M., van Mil, F. N., Ivanyi, D., and Ramaekers, F. C. (1992). Immunohistochemistry with Keratin monoclonal antibodies in canine tissues: urogenital tract, respiratory tract, (neuro-) endocrine tissues, choroid plexus and spinal cord, J Vet Med 39, 721-40.
7. Vos, J. H., van den Ingh, T. S., Ramaekers, F. C., Molenbeek, R. F., de Neijs, M., van Mil, F. N., and Ivanyi, D. (1993). The expression of Keratins, vimentin, neurofilament proteins, smooth muscle actin, neuron-specific enolase, and synaptophysin in tumors of the specific glands in the canine anal region, Vet Pathol 30, 352-61.
8. Verhagen, A. P., Ramaekers, F. C., Aalders, T. W., Schaafsma, H. E., Debruyne, F. M., and Schalken, J. A. (1992). Colocalization of basal and luminal cell-type Cytokeratins in Human prostate cancer, Cancer Res 52, 6182-7.
9. van Leenders, G., Dijkman, H., Hulsbergen-van de Kaa, C., Ruiter, D., and Schalken, J. (2000). Demonstration of intermediate cells during Human prostate epithelial differentiation in situ and in vitro using triple-staining confocal scanning microscopy, Lab Invest 80, 1251-8.
Protein Reference(s)
Database Name: UniProt
Accession Number: multiple
Safety Datasheet(s) for this product:
| NM_Sodium Azide |
|
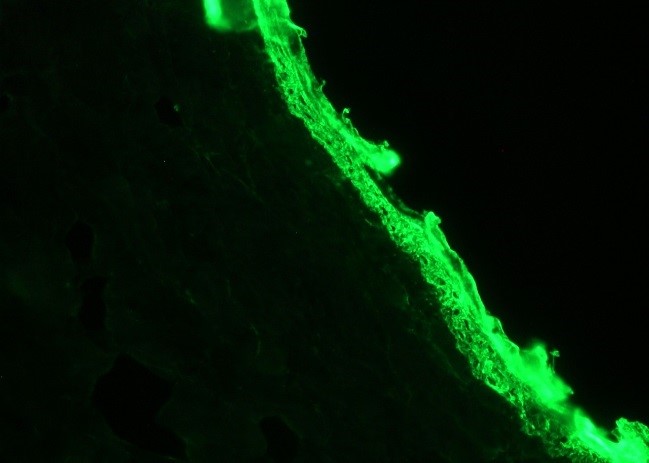
Figure 1. Indirect immunofluorescence staining of frozen section of dog skin with MUB0312P (RCK103) showing positive staining in basal cells of epidermis. Dilution 1:100. |

Figure 1. Indirect immunofluorescence staining of frozen section of dog skin with MUB0312P (RCK103) showing positive staining in basal cells of epidermis. Dilution 1:100.
